Product
Evaluation
We conduct product evaluations
based on scientific evidence
for medical, pharmaceutical,
and health-related products.
Our fundamental approach is to conduct objective and systematic evaluations from multiple perspectives including efficacy, safety, and reproducibility, independent of product positioning or intended use.
Key Evaluation
Perspectives
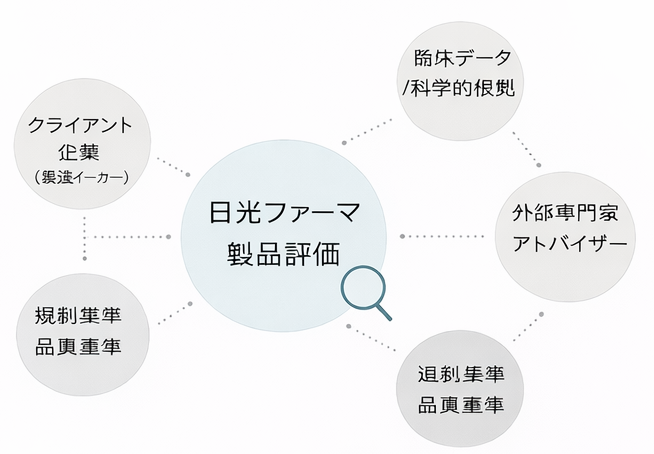
Nikko Pharma's product evaluation is structured based on the following perspectives:
Scientific Validity
Examination of validity based on existing literature, research data, and submitted materials
Safety Assessment
Confirmation of ingredient characteristics, usage conditions, and known risk information
Reproducibility & Consistency
Verification of reproducibility and consistency in data and design
Product Design Rationality
Examination of scientific rationality in formulation design and component composition
Appropriateness of Information Expression
Confirmation that product information and descriptions are consistent with scientific evidence
Key
Deliverables

Evaluation results are organized and provided to clients in the following formats:
Product Evaluation Report
Organization and findings from a scientific perspective
Clarification of evaluation scope and prerequisites
Issue organization for future considerations (as needed)
*Evaluation content does not imply specific approval, guarantee, or recommendation.
Nikko's
Role
Nikko Pharma places importance on our responsibility as "evaluators" from a position independent of product developers and sellers.
Our role is to conduct evaluations based on scientific evidence without bias toward any particular position, and to objectively organize product characteristics.
Contact
Inquiries about Product Evaluation
For inquiries about product evaluation, please feel free to contact us.
→ View More (Contact Us)